Vision Vices – Focus on your eye health
Be honest now: are you guilty of any of these? We've ranked the nation's most cut corners when it comes to building a good eye care routine and share easy-to-implement tips to improve your eyesight. Read More

Be honest now: are you guilty of any of these? We've ranked the nation's most cut corners when it comes to building a good eye care routine and share easy-to-implement tips to improve your eyesight. Read More

Festival season is now just around the corner. So if you’re getting ready to head out into the fields this summer, it’s worth making sure you have everything you need to make the most of your experience. Read More

Blinking is as natural as breathing but our emotions and intentions can alter our blinking pattern. Discover what your blinking habits reveal about you. Read More

Do you also love winter? A wonderful season to take it easy or to get out and get fit! If you wear contact lenses, you don't want to be bothered by your eyes or your lenses during this winter. So read on for our tips and enjoy this season to the fullest. Read More

Is love at first sight real? Whether you’re a believer or a sceptic, the eyes do play a big role when it comes to falling in love. Read More
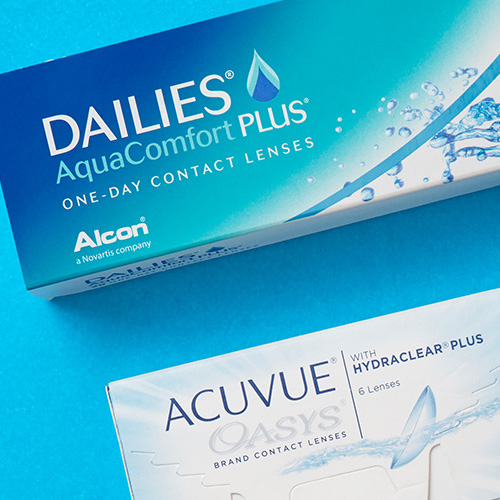
Check out what contact lenses, eye drops and solutions our customers are loving, and embrace comfort and a clearer vision for the rest of the year! Read More

Wearing contact lenses is very common these days. Many people are happy that they can experience the convenience of lenses every day. But when were lenses actually invented and by whom? Read on to find out. Read More

Buying your contact lenses in bulk is the way to go, as you’ll save yourself money and prevent any panic by never running out. Plus, you can enjoy peace of mind with our 365-day guarantee and help decrease our carbon footprint. Read More

Discover the best lenses to see you through the hectic final months of 2022 and start 2023 on the right foot with this blog. Read More

Long-time Vision Direct customer Julia shares her daily routine with us and explains how her contact lenses enable her to live a busy, fulfilling life. Read More